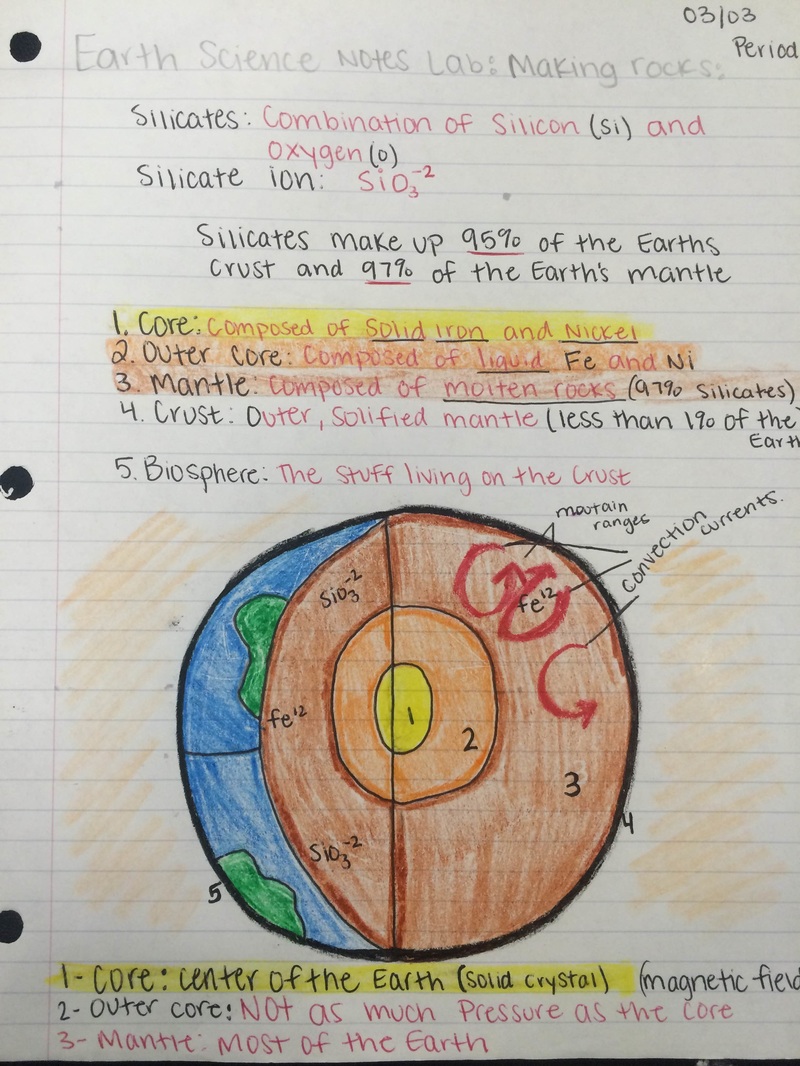
Background Knowledge:
Making Cobalt Silicate:
My Experience!
Looking Back:
In this experiment, we made rocks! We had a vial filled with sodium silicate, a clear liquid. We then filled very little Cobalt Silicate into the vial tube. We then added another chemical and watched the rocks slowly grow. The experiment worked out just not how we wanted. I wish it could've been a little bigger. Other than that it was really cool watching rocks we made grow!
Looking Beyond:
We started with cobalt chloride and sodium silicate. We put both of them together into a solution. We got one molecule that has chlorine and one molecule that has sodium. Every element wants a shell of eight. So sodium and chlorine are extremely reactive to each other because sodium has one outer most electron and chlorine has 7. When they bond chlorine takes that one valence electron and now they both have full shells. So now chlorine is negative because it has a extra electron and sodium is now positive because it is missing a electron. Now, because of the difference in charge they bond. It's like positive and negative magnets, they bond together. So when they bonded together it created a salt. At this point the Cobalt and the Chloride broke away from each other So when they sat in the solution the silicate and Cobalt bonded, creating Cobalt Silicate which is the rock that we grew. I didn't have very many questions. I was more concerned about what would happen if I touched it...
Looking Inward:
I feel like the results we got were okay and that they came out the way they were supposed to. I didn't really participate in the putting the rocks into the tube. I really only took pictures which I think is still a big deal. I was safe around the chemicals and I followed all the instructions. So I would have to say I did pretty good.
Looking Outward:
I worked with Kim, and Omar. Our group performed rather well. We all did our part of picture taking and we all followed the safety rules and guidelines. It was kind of fun working with them for the first time in a while. I forgot how good we work together.
Looking Forward:
If I could do this experiment again, I would use a larger amount of both substances and make bigger rocks to get a better look at the results. If I were to choose a group for next time, I would choose the same people, Kim, Gloriann and Omar. We all work really good together and share the responsibility. To continue this experiment I would want to try different combinations of elements and see which ones combine and make rocks and which ones don't.
Take Aways:
What I learned from this was how to make rocks. It's more simple than you think. I also learned that when making rocks you're not supposed to shake the tube or else the rocks inside will break. In this experiment I learned about cobalt chloride and sodium silicate and how they bond! Not going to lie I kind of feel smart! :)
Looking Back:
In this experiment, we made rocks! We had a vial filled with sodium silicate, a clear liquid. We then filled very little Cobalt Silicate into the vial tube. We then added another chemical and watched the rocks slowly grow. The experiment worked out just not how we wanted. I wish it could've been a little bigger. Other than that it was really cool watching rocks we made grow!
Looking Beyond:
We started with cobalt chloride and sodium silicate. We put both of them together into a solution. We got one molecule that has chlorine and one molecule that has sodium. Every element wants a shell of eight. So sodium and chlorine are extremely reactive to each other because sodium has one outer most electron and chlorine has 7. When they bond chlorine takes that one valence electron and now they both have full shells. So now chlorine is negative because it has a extra electron and sodium is now positive because it is missing a electron. Now, because of the difference in charge they bond. It's like positive and negative magnets, they bond together. So when they bonded together it created a salt. At this point the Cobalt and the Chloride broke away from each other So when they sat in the solution the silicate and Cobalt bonded, creating Cobalt Silicate which is the rock that we grew. I didn't have very many questions. I was more concerned about what would happen if I touched it...
Looking Inward:
I feel like the results we got were okay and that they came out the way they were supposed to. I didn't really participate in the putting the rocks into the tube. I really only took pictures which I think is still a big deal. I was safe around the chemicals and I followed all the instructions. So I would have to say I did pretty good.
Looking Outward:
I worked with Kim, and Omar. Our group performed rather well. We all did our part of picture taking and we all followed the safety rules and guidelines. It was kind of fun working with them for the first time in a while. I forgot how good we work together.
Looking Forward:
If I could do this experiment again, I would use a larger amount of both substances and make bigger rocks to get a better look at the results. If I were to choose a group for next time, I would choose the same people, Kim, Gloriann and Omar. We all work really good together and share the responsibility. To continue this experiment I would want to try different combinations of elements and see which ones combine and make rocks and which ones don't.
Take Aways:
What I learned from this was how to make rocks. It's more simple than you think. I also learned that when making rocks you're not supposed to shake the tube or else the rocks inside will break. In this experiment I learned about cobalt chloride and sodium silicate and how they bond! Not going to lie I kind of feel smart! :)
| sodium-silicate-n.pdf |